HEAT IN REFRIGERATION
The laws of thermodynamics can help us to understand what heat is all about. The first law of thermodynamics states that energy can be neither created nor destroyed, but can be converted from one form to another. This means that most of the heat the world experiences is not being continuously created but is being converted from other forms of energy, like fossil fuels (gas and oil). This heat can also be accounted for when it is transferred from one substance to another. Temperature describes the level of heat with reference to absolute zero, or the temperature at which there is no heat present in a substance. The term used to describe the quantity of heat or heat content is known as the British thermal unit (Btu), which indicates how much heat is contained in a substance. The rate of heat transfer can be determined by considering the time it takes to transfer a certain amount of heat energy. Air-conditioning and heating equipment is rated in Btu/h, where the “h” represents “hour.” An air conditioning system that is rated at 24,000 Btu/h has the capacity to remove 24,000 Btu of heat energy from the structure every hour.
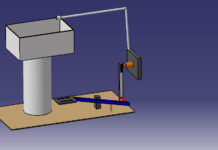
The Btu is defined as the amount of heat required to raise the temperature of 1 pound (lb) of water 1°F. For example, when 1 lb of water (about 1 pint) is heated from 68°F to 69°F, 1 Btu of heat energy is absorbed into the water, Figure 1. When a temperature difference exists between two substances, heat transfer will occur. The next three sections in this unit discuss the three types of heat transfer, namely, conduction, convection, and radiation.

Temperature difference is the driving force behind heat transfer. The greater the temperature difference, the greater the heat transfer rate. Heat flows naturally from a warmer substance to a cooler substance. Rapidly moving molecules in the warmer substance give up some of their energy to the slower-moving molecules in the cooler substance. The warmer substance cools because the molecules have slowed. The cooler substance becomes warmer because the molecules are moving faster. So, the next time you pour warm soda into a glass filled with ice, remember that the ice isn’t making the soda cold, the soda is making the ice hot! The following example illustrates the difference in the quantity of heat compared with the level of heat. One tank of water weighing 10 lb (slightly more than 1 gallon [gal]) is heated to a temperature level of 200°F. A second tank of water weighing 100,000 lb (slightly more than 12,000 gal) is also heated to 200°F. The 10-lb tank will cool to room temperature much faster than the 100,000-lb tank because even though the temperature of the water in both tanks is the same, there is much more heat contained in the larger tank, Figure 2.
A comparison using water may be helpful in differentiating between the heat intensity level and the quantity of heat. A 200-ft-deep well would not contain nearly as much water as a large lake with a water depth of 25 ft. The depth of water (in feet) tells us the level of water, but it in no way expresses the quantity (gallons) of water. In practical terms, each piece of heating equipment is rated according to the amount of heat it will produce in a given time period. If the equipment had no such rating, it would be difficult for a buyer to intelligently choose the correct appliance. The rating of a gas or oil furnace used to heat a home is permanently printed on a nameplate. Either furnace would be rated in Btu per hour, which is a rate at which heat energy is transferred into the structure. For now, it is sufficient to say that if a 75,000-Btu/h furnace is needed to heat a house on the coldest day, one should choose a furnace rated at 75,000 Btu/h. If a smaller furnace is chosen, the house will begin to get cold any time the heat loss of the house exceeds the furnace output in Btu/h.

To describe the absence of heat, the term cold is often used as a comparative term to describe lower temperature levels. Because all heat is a positive value in relation to no heat, cold is not a true value; it is really an expression of comparison. When a person says it is cold outside, the term is being used to express a relationship to the normal expected temperature for the time of year or to the inside temperature. Cold has no numerical value and is used by most people as a basis of comparison only. Cold is sometimes referred to as a “level of heat absence.”
Copied from REFRIGERATION & AIR CONDITIONING TECHNOLOGY by WILLIAM C. WHITMAN, WILLIAM M. JOHNSON, JOHN A. TOMCZYK and EUGENE SILBERSTEIN